Hello |안녕하세요 | Hola | 你好 | Olá |नमस्ते | Hallo | வணக்கம் | Bonjour
Welcome to the Bishai Laboratory

Tuberculosis:
Tuberculosis (TB) is an ancient and complex human illness that we now know exists as a disease spectrum resulting from a dynamic interaction between the host immune system and the bacilli. Aside from COVID-19, TB is the leading cause of global morbidity and mortality from a single infectious disease agent. Mycobacterium tuberculosis (Mtb), the etiological agent of TB disease in humans, latently infects approximately one-fourth of the world’s population and kills approximately 1.5 million individuals a year.
Recent Publication(s):
FRESH OFF THE PRESS: Special free pre-view available till Sept 1, 2022!
Journal: Trends in Immunology (Cell Press)
Issue: 2022 TBD (FEATURE REVIEW)
PMID: TBD
Link: https://www.sciencedirect.com/science/article/pii/S147149062200120X?via%3Dihub
Title: Genetic and hormonal mechanisms underlying sex-specific immune responses in tuberculosis
Authors: Manish Gupta, Geetha Srikrishna, Sabra L. Klein, and William R. Bishai
Abstract:
Tuberculosis (TB), the world’s deadliest bacterial infection, afflicts more human males than females, with a male/female (M/F) ratio of 1.7. Sex disparities in TB prevalence, pathophysiology, and clinical manifestations are widely reported, but the underlying biological mechanisms remain largely undefined. This review assesses epidemiological data on sex disparity in TB, as well as possible underlying hormonal and genetic mechanisms that might differentially modulate innate and adaptive immune responses in males and females, leading to sex differences in disease susceptibility. We consider whether this sex disparity can be extended to the efficacy of vaccines and discuss novel animal models which may offer mechanistic insights. A better understanding of the biological factors underpinning sex-related immune responses in TB may enable sex-specific personalized therapies for TB.
————————————————————————————————–
Journal: eLife
Issue: 2022 Apr 27;11:e73055. doi: 10.7554/eLife.73055.
PMID: 35475970
Title: Allosteric cooperation in β-lactam binding to a non-classical transpeptidase
Authors: Ahmad N, Dugad S, Chauhan V, Ahmed S, Sharma K, Kachhap S, Zaidi R, Bishai WR, Lamichhane G, Kumar P.
Abstract:
L,D-transpeptidase function predominates in atypical 3 → 3 transpeptide networking of peptidoglycan (PG) layer in Mycobacterium tuberculosis. Prior studies of L,D-transpeptidases have identified only the catalytic site that binds to peptide moiety of the PG substrate or β-lactam antibiotics. This insight was leveraged to develop mechanism of its activity and inhibition by β-lactams. Here, we report identification of an allosteric site at a distance of 21 Å from the catalytic site that binds the sugar moiety of PG substrates (hereafter referred to as the S-pocket). This site also binds a second β-lactam molecule and influences binding at the catalytic site. We provide evidence that two β-lactam molecules bind co-operatively to this enzyme, one non-covalently at the S-pocket and one covalently at the catalytic site. This dual β-lactam-binding phenomenon is previously unknown and is an observation that may offer novel approaches for the structure-based design of new drugs against M. tuberculosis.
————————————————————————————————–
Journal: Nature Communication
Issue: Nat Commun. 2022 Feb 15;13(1):878. doi: 10.1038/s41467-022-28509-z.
PMID: 35169141
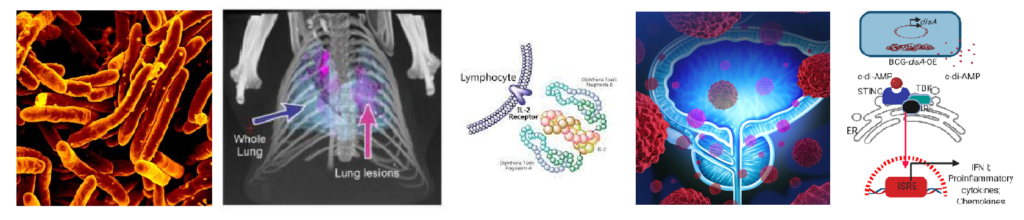
Title: Re-engineered BCG overexpressing cyclic di-AMP augments trained immunity and exhibits improved efficacy against bladder cancer.
Authors: Singh AK, Praharaj M, Lombardo KA, Yoshida T, Matoso A, Baras AS, Zhao L, Srikrishna G, Huang J, Prasad P, Powell JD, Kates M, McConkey D, Pardoll DM, Bishai WR, Bivalacqua TJ.
Abstract
In addition to its role as a TB vaccine, BCG has been shown to elicit heterologous protection against many other pathogens including viruses through a process termed trained immunity. Despite its potential as a broadly protective vaccine, little has been done to determine if BCG-mediated trained immunity levels can be optimized. Here we re-engineer BCG to express high levels of c-di-AMP, a PAMP recognized by stimulator of interferon genes (STING). We find that BCG overexpressing c-di-AMP elicits more potent signatures of trained immunity including higher pro-inflammatory cytokine responses, greater myeloid cell reprogramming toward inflammatory and activated states, and enhances epigenetic and metabolomic changes. In a model of bladder cancer, we also show that re-engineered BCG induces trained immunity and improved functionality. These results indicate that trained immunity levels and antitumor efficacy may be increased by modifying BCG to express higher levels of key PAMP molecules.
Like and Follow: